Clock Foundation’s GrimAge Biological Age Test Review
Epigenetic age test provides information beyond biological age
Join the club for FREE to access the whole archive and other member benefits.
A research paper released last week is causing a stir in the ageing research community. It announces the successful rejuvenation of laboratory rats – by simple injection of blood plasma fraction - reducing their equivalent human age of 80 years old to only 40 years old.
If this was even only partially replicated in humans (let’s say a reduction of 10% instead of 50%) this would be a society changing therapy. Instead of treating one disease at a time, plasma fraction treatment could significantly delay the onset of many age-related diseases at the same time.
This has not been written by some crackpot scientist, or an over-eager post-grad. Harold Katcher has decades of experience in academia. His previous work has been cited thousands of times and he was part of the team behind the discovery of the human breast cancer gene, BRCA1, and teaches online as a professor for the University of Maryland.
Note, bioRxiv is a preprint server which means the work has not yet been peer-reviewed and formally published in an academic journal. However, there are some highly-regarded researchers involved in this work (including Steve Horvath, professor at UCLA with an ageing clock named after him) and other leading ageing academics (such as Harvard professor David Sinclair) who have evaluated the published data and have given positive feedback.
By injecting old rats with components of young rats’ blood plasma, the researchers observed a wide range of improvements in their physical state – including epigenetic clocks, blood biomarkers, physical strength, and mental performance – almost everything that declines with age.
Given the title of the research paper focused on “measurement of epigenetic age” I was initially sceptical that the treatment might have been so fine tuned to make the biggest impact on their epigenetic clocks that there was little, no, or even a negative effect, on the body as a whole.
However, reading through the paper, it is clear that the impact is universally positive, and most changes are significant (not like most pharmaceutical drugs that struggle to beat the placebo effect). The overall reduction in epigenetic age (considered one of the most accurate measures of biological age) was 54%.
Briefly, epigenetic clocks are a way of measuring the biological age of mammals. They look at how DNA is being expressed (read and used) in cells. Remember that every cell in your body contains the same DNA (with all the instructions, i.e. genes, needed to build and maintain a whole new body), but different cell types (skin, blood, liver, etc.) behave as needed in their tissue because the relevant part of the DNA is active. As we age, this DNA methylation (DNAm) starts to go awry, resulting in cells malfunctioning and therefore tissues and organs, that are composed of them, also starting to fail. Epigenetics is the study of this gene expression, and epigenetic clocks can track how much damage has been done, and therefore how old the cell is.
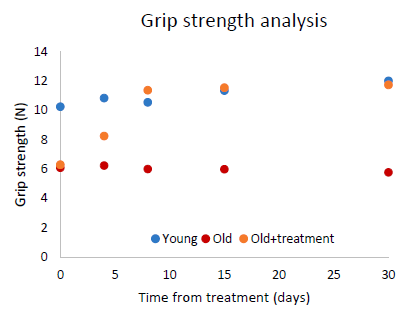
Typically, the biomarkers of the old rats trended towards that of the younger rats. Sometimes this changed happened in days or weeks, for lots of biomarkers it took months, but most ended up being the same (or almost the same) as that of rats half of their age (the young controls). For example, below is the grip strength of the rats. The blue dots represent the strength of young rats, which hovers around 10-12 Newtons (a measurement of force). Old rats (orange dots) initially had a grip strength of 6 N, with the untreated old rats (red dots) remaining at that level. But within 1-2 weeks of treatment, the test rats increased their strength to the same level as the young ones.
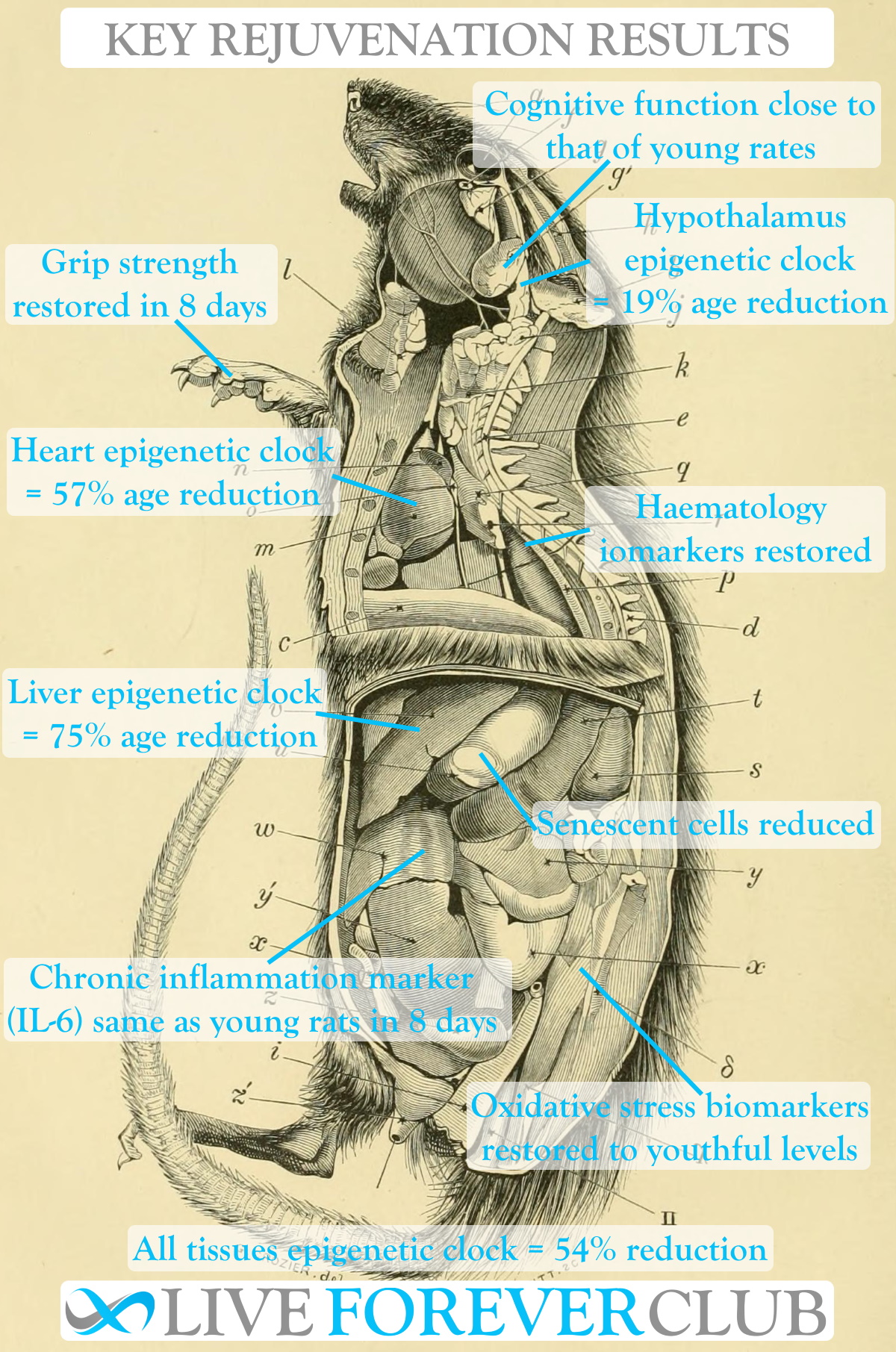
Here is a summary of all of the characteristics that were measured and how they changed after the plasma fraction treatment.
| Biomarker | Result | Health impact |
|---|---|---|
| BIOLOGICAL AGE | ||
| Liver epigenetic clock | 75% reduction | |
| Blood epigenetic clock | 66% reduction | |
| Heart epigenetic clock | 57% reduction | |
| Hypothalamus epigenetic clock | 19% reduction | |
| All tissues epigenetic clock | 54% reduction | |
| HAEMATOLOGY | ||
| Haemoglobin level (Hb) | Increased to youthful levels within 150 days | Measure of ability to carry oxygen around the body |
| Mean corpuscular volume (MCV) | Decreased to youthful levels within 150 days | Size of the red blood cells |
| Mean corpuscular haemoglobin (MCH) | Indistinguishable from youthful levels within 60 days | Amount of haemoglobin in an average red blood cell |
| Mean corpuscular haemoglobin concentration (MCHC) | Indistinguishable from youthful levels within 60 days | Concentration of haemoglobin in an average red blood cell |
| Hematocrit (HCT) level | Decreased to youthful levels within 60 days, but rising more rapidly again at 150 days | Volume red blood cells take up in the blood |
| Red blood cells count (RBC) | Close to youthful levels within 150 days | |
| Platelets | Only a very small reduction observed | |
| White blood cells | Indistinguishable from youthful levels within 60 days | Key to the body’s immune system |
| Lymphocytes | Close to youthful levels within 150 days | |
| BIOCHEMISTRY | ||
| Bilirubin (total and direct) | Reduced to close to youthful levels within 150 days | Lower level suggests improved liver function |
| Serum glutamic-pyruvic transaminase (SGPT) | Reduced to youthful levels within 150 days | High levels can indicate liver damage or injury |
| Serum glutamic-oxaloacetic transaminase (SGOT) | Reduced to youthful levels within 150 days | High levels can indicate liver damage or injury |
| Triglycerides (TG) | Reduced to youthful levels within 150 days | High levels can indicate cirrhosis, diabetes or pancreatitis |
| HDL | increased to close to youthful levels within 150 days | Low levels can mean a higher risk of heart disease |
| Cholesterol | Reduced to close to youthful levels within 150 days | High levels can mean a higher risk of heart disease |
| Glucose | Reduced to youthful levels within 150 days | High levels can indicate diabetes or pancreatitis |
| Creatinine | Reduced to youthful levels within 150 days | High levels can indicate kidney damage |
| Blood urea nitrogen (BUN) | More than halfway to youthful levels within 150 days | High levels can indicate kidney damage |
| Total protein | Reduced to close to youthful levels within 150 days | High levels can indicate chronic infection or inflammation |
| OXIDATIVE STRESS and INFLAMMATION | ||
| Malondialdehyde (MDA) | Reduced to almost the same as young rats in all organs | Indicator of oxidative stress in cells and tissues |
| Glutathione (GSH) | Increased to almost the same as young rats in all organs | Anti-oxidant produced naturally by the liver |
| Catalase | Increased to almost the same as young rats in brain, heart and liver. Halfway improvement in liver. | Enzyme protects the cell from oxidative damage by reactive oxygen species (ROS) |
| Superoxide Dismutase | Increased to almost the same as young rats in all organs | Breaks down superoxide radicals to prevent cell damage |
| Interleukin 6 (IL-6) | Reduced to young rat levels within 8 days | Elevated concentrations indicate chronic inflammation |
| Tumor necrosis factor α (TNF-α) | Approximately two-third reduction in difference between old and young levels within 8 days | High levels can indicate chronic inflammatory disorders, such as rheumatoid arthritis and inflammatory bowel disease |
| Nrf2 | Increased to be indistinguishable from youthful levels at end of experiment | Modulates inflammation and oxidative stress |
| Senescent cells (SA-β-galactosidase staining) | Significant reduction in brain tissue. Almost youthful levels in liver tissue. Low rate retained in heart and lung tissue. | Senescent cells are an anti-ageing target in their own right (using senolytics) |
| PHYSICAL | ||
| Weight | Very slight reduction | |
| Grip strength | Indistinguishable from youthful levels within 8 days | |
| COGNITIVE FUNCTION | ||
| Barnes maze latency | Latency on first day of training reduced halfway to young rats within 120 days. Latency at end of 6-day training period reduced to almost the same as young rats within 56 days. | Demonstrates improved spatial learning and memory |
As comprehensive as this list of positive outcomes is, equally important is the question of adverse side-effects. It would be no benefit if all of these biomarkers were improved, but the rats had a greatly increased chances of cancer (a common risk with anti-ageing treatments) or suffered from catastrophic organ failure.
The good news is that the paper’s authors report no overt signs of physical or behavioural abnormality in the tested rats. Their appetite was not affected, and post experiment examinations of various organs (lungs, kidney, heart, spleen, brain, testes) did not indicate any abnormalities. Some mild granular degeneration of liver was observed but only at the same level as in the young controls.
6 old rats (109 weeks old) were injected with a plasma fraction at the start of the experiment and then, again, 95 days later. To study the effect of the treatment, many factors (as described in the table above) were analysed at regular intervals and compared to two other groups of rats: (i) 6 old rats – the control group, and (ii) 6 young rats (30 weeks old). Fortunately, you don’t have to worry about the placebo affect with murine models.
This is the equivalent of giving the injection to people who are 68, and then again at 76. And then, when chronologically aged 81, checking their biological age to find that it is the same as a 40-year-old.
Let’s start with what plasma is. Blood plasma is, primarily, the liquid (mainly water) that red and white blood cells are transported around the body in. However, it also contains many important components of its own – including minerals, nutrients, and proteins (6-8%).
There are thousands of different types of proteins in the blood, ranging in concentration from picograms to milligrams per millilitre – that’s a difference of a million-million-fold, which makes analysing what all them do a significant challenge. The most common proteins are albumins, globulins and fibrinogens.
Importantly for this research, many proteins in blood plasma are signalling molecules (such as cytokines and hormones) that enable cells to communicate and interact with each other at long distances, and for the body to regulate itself. The authors of this paper believe that aging is coordinated, and therefore can be systemically controlled, through the circulatory system. This is contrary to others’ views (including SENS) that ageing is a random process that impacts different organs independently.
A plasma fraction is simply a subset of the whole blood plasma. Blood plasma fractionation is a common process used to separate the various components of the plasma. What we don’t know at this stage, is just which components (what proteins and other molecules) was included in the plasma fraction injected into the old rats. This is understandable as this is commercially sensitive – the authors have already setup a company, Nugenics Research, to take this treatment to human clinical trials.
Katcher et al are not the only ones taking this approach. Jesse Karmazin formed Ambrosia in 2016 and ran a paid trial providing transfusions of blood plasma from under-25s, and claims improvements in biomarkers related to Alzheimer's disease, cancer, and inflammation. Taking that a step further, other companies have joined the race to identify which factors in the blood really help. Alkahest are focussed on two groups of functional plasma proteins, one trying to improve Alzheimer’s and Parkinson’s diseases, the other focussed on wet age-related macular degeneration.
This is the most significant rejuvenation seen in a mammal so far. Obviously, it needs to be verified and reproduced, but the results are so dramatic that it seems unlikely that this, or a similar treatment will not work in some way. Though it’s worth reminding ourselves that many potential therapies work in the lab but not in humans – however with such a wide range of physiological improvements its hard to imagine them all falling at the last hurdle.
The big unanswered question is, what would have been the impact on the average and maximum lifespan of the rats if they weren’t sacrificed at 155 days to perform biopsies of their organs? Would they have lived longer, and how much longer if the treatment was repeated every 95 days? Worst case, there would be no impact, but with all of the improvements to the biomarkers the equivalent treatment in humans would likely result in a much extended healthspan – compacting all age related afflictions into the last few months of life, which wouldn’t be a bad thing.
We’ll have to wait for the announcement by Nugenics Research (founded by Harold Katcher and Akshay Sanghavi)) as to their plans to commercialize the "Elixir" treatment. Ambrosia struggled to get enough people to pay to participate in their trial of young (unfractioned) plasma, but it’s likely that Nugenics will raise enough investment to fund a human clinical trial, and I doubt they will have any shortage of volunteers.
It will also be interesting to find out whether this elixir can be mass produced, by growing the molecules in a lab, or whether it will depend on a steady supply of teenagers being paid for the youthful plasma.
Of course, long-term there could be some nasty surprises (cancer or other cellular disruption) but that may be a risk worth taking if your days are already numbered, and in the time bought with a plasma fraction treatment who knows what cures will be discovered for other conditions.
All things considered, this is a sensational piece of meticulously performed research that no doubt will raise the expectations of everyone interest in rejuvenation, life extension, or even just a longer healthspan.
Predictions of curing human ageing in the next 20 years have definitely become a lot more reasonable with these results.
Reversing age: dual species measurement of epigenetic age with a single clock – bioRxiv
Harold Katcher profile – ResearchGate
Click on resource name for more details.
Summary of London Futurists’ Engineering Greater Human Resilience Panel Discussion
NMN Supplements Personal Case Study
Epigenetic age test provides information beyond biological age
Survival curve shows increase in average lifespan, and now Sima has become the longest-lived Sprague Dawley rat
Adrian tries out this home epigenetic test and is impressed with the level of detail in the reports
How does the cheapest epigenetic age test on the market compare?